The first-ever year-long systemic flea and tick protection for dogs

A revolutionary injectable with a trusted ingredient: fluralaner

Available exclusively through vets



A VET-EXCLUSIVE
Designed to be the ultimate support for you and your efforts to control parasite infestations and vector-borne diseases.
CERTAINTY
Administering the injection in practice ensures no gaps in year-long protection, eliminating reliance on pet owners to administer products correctly, at the proper frequency.
CONVENIENCE
BRAVECTO injectable is administered as a single injection during a vet visit to provide year round protection.
COMFORT
The innovative suspension of fluralaner crystals allows for simple administration and a favorable pet and owner experience.


KEEP PATIENTS PROTECTED FOR A FULL YEAR
BRAVECTO injectable
protects dogs 6 months of age or older against multiple species of fleas and tick, including:

Ctenocephalides felis
Ctenocephalides canis

Ixodes ricinus
Ixodes hexagonus
Dermacentor reticulatus
Rhipicephalus sanguineus
BRAVECTO injectable delivers year-long protection
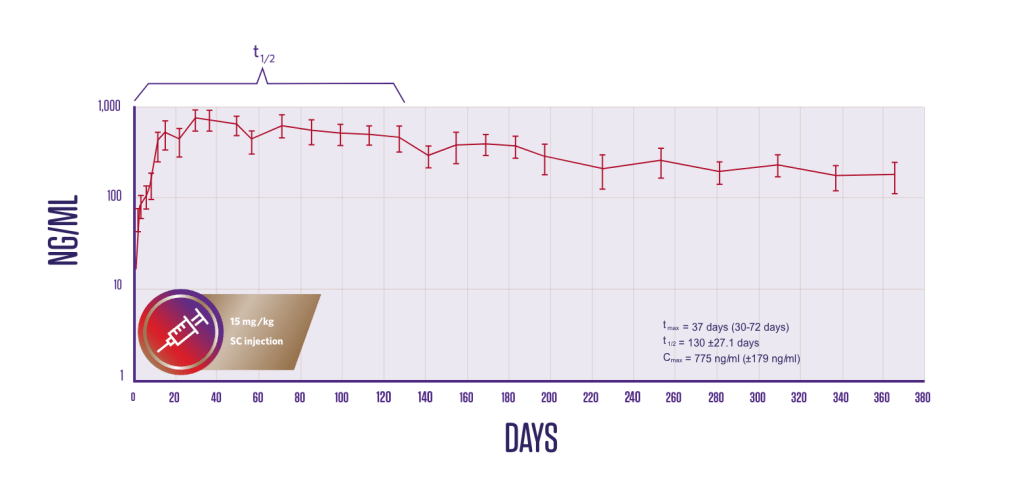
Pharmacokinetics
PHARMACOKINETICS
Plasma fluralaner concentrations following a single subcutaneous administration1
Flea efficacy

IMMEDIATE AND PERSISTENT FLEA-KILLING ACTIVITY FOR 12 MONTHS
Duration of efficacy against Ctenophalides felis – adulticide2
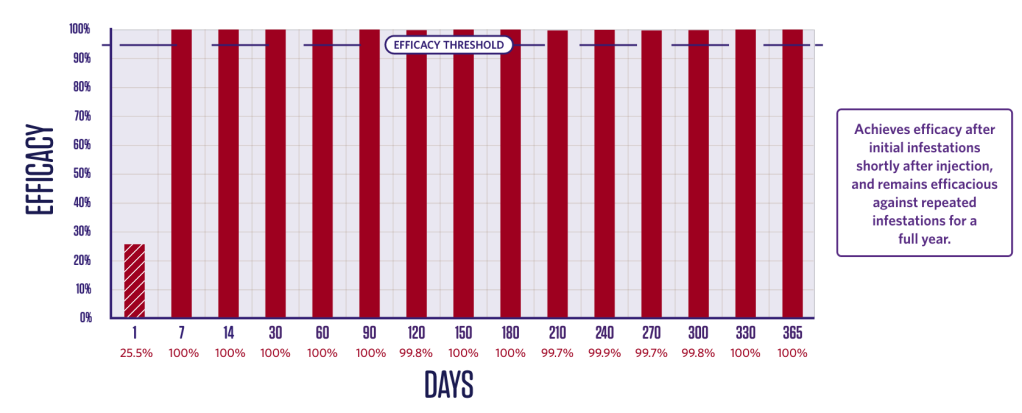
Newly-infested fleas are killed within 24 hours from one week to 12 months after treatment.2,3

2 groups (1 treated, 1 control);
10 dogs in each group.

Dogs were infested with 100 fleas on Day -1 and treated on Day 0. Efficacy against the initial infestation was evaluated on Day 1.

Persistent efficacy evaluated at
intervals from Day 7 to Day 366
following repeated re-infestations.
*Flea counts are arithmetic means. Percent effectiveness is based on arithmetic means.

NEWLY EMERGED FLEAS ARE KILLED BEFORE VIABLE EGGS ARE PRODUCED
Efficacy against C. felis – egg production.2
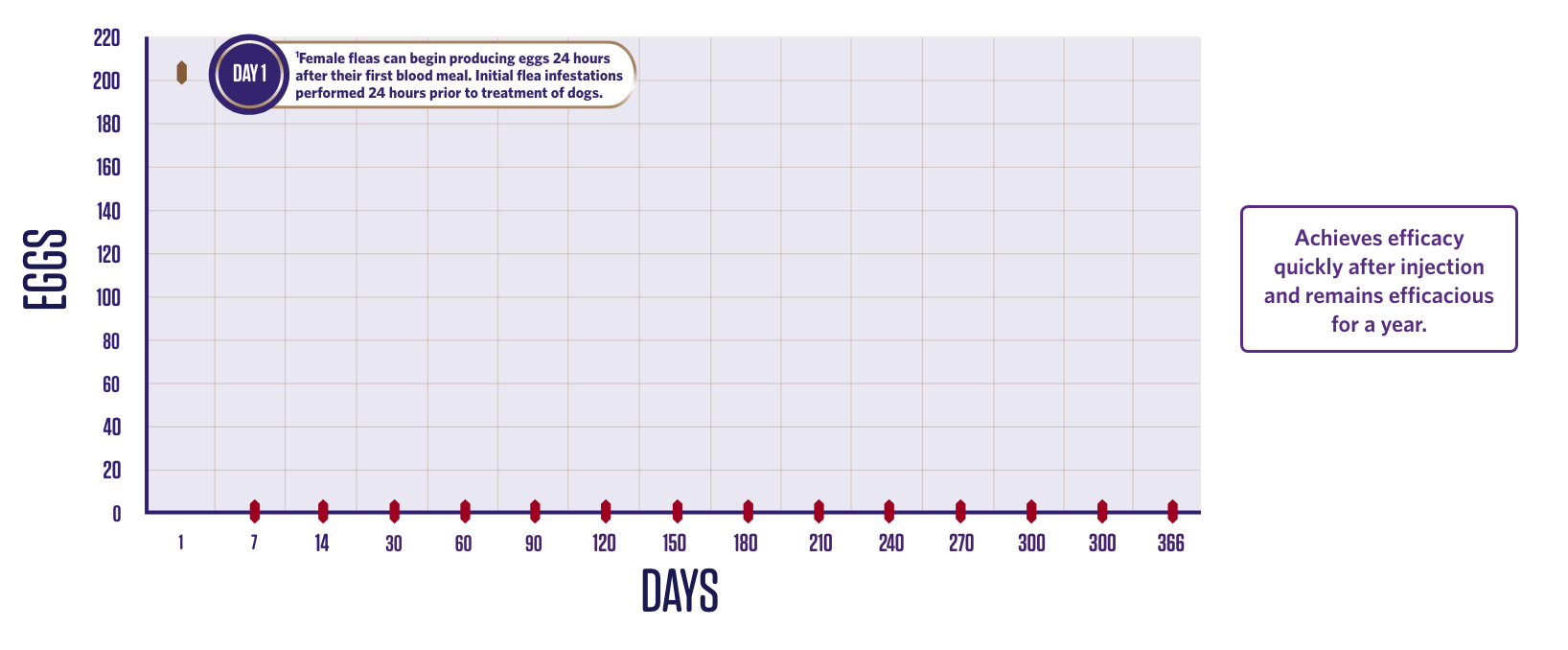
The flea life cycle is broken due to the rapid onset of action and long-lasting efficacy against adult fleas on the animal and the absence of viable egg production.3

2 groups (1 treated, 1 control);
10 dogs in each group.

Dogs were infested with 100 fleas on Day -1 and treated on Day 0. Efficacy against the initial infestation was evaluated on Day 1.

Persistent efficacy evaluated at
intervals from Day 7 to Day 366
following repeated re-infestations.
*Flea egg counts are arithmetic means.
Tick efficacy

PERSISTENT TICK KILLING ACTIVITY FOR 12 MONTHS
Duration of efficacy against Rhipicephalus sanguineus4
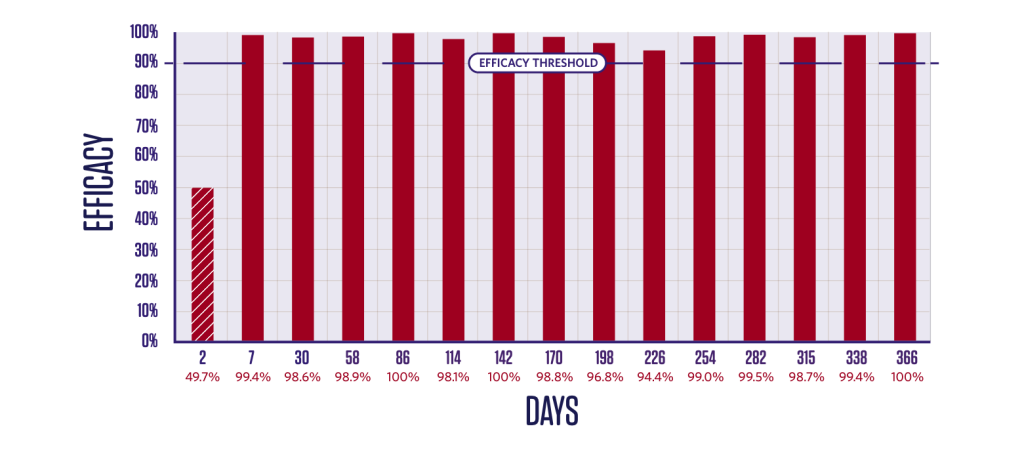
Persistent Rhipicephalus sanguineus tick-killing activity from Day 4 after treatment for 12 months. Newly-infested R. sanguineus ticks are killed.3,4

2 groups (1 treated, 1 control);
10 dogs in each group.

Dogs were infested with 50 ticks on Day -2 and treated on Day 0. Efficacy against the initial infestation was evaluated on Day 2.

Persistent efficacy evaluated at intervals from Day 7 to Day 366 following repeated re-infestations.
*Tick counts are arithmetic means.

PERSISTENT TICK KILLING ACTIVITY FOR 12 MONTHS
Duration of efficacy against Dermacentor reticulatus5
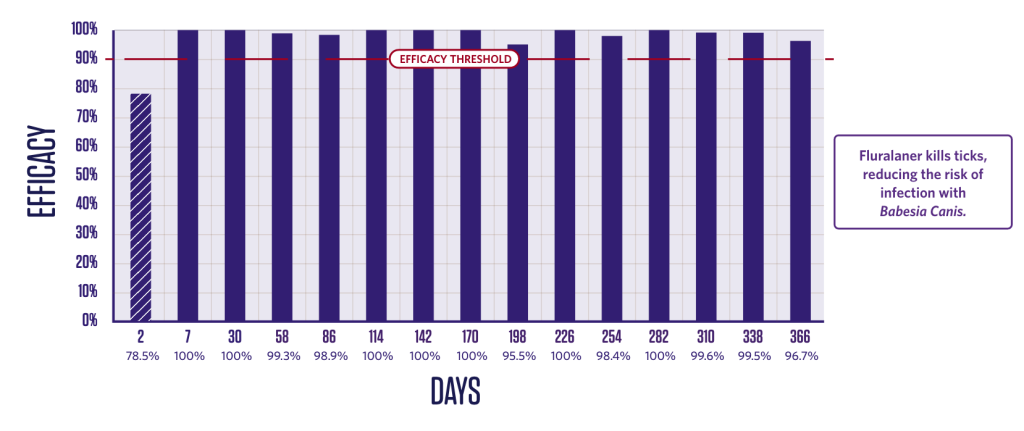
Persistent Dermacentor reticulatus killing activity from Day 3.
Newly-infested D. reticulatus ticks are killed.3,5

2 groups (1 treated, 1 control);
10 dogs in each group.

Dogs were infested with 50 ticks on Day -2 and treated on Day 0. Efficacy against the initial infestation was evaluated on Day 2.

Persistent efficacy evaluated at intervals from Day 7 to Day 366 following repeated re-infestations.
*Tick counts are arithmetic means.

PERSISTENT TICK KILLING ACTIVITY FOR 12 MONTHS
Duration of efficacy against Ixodes ricinus6,7
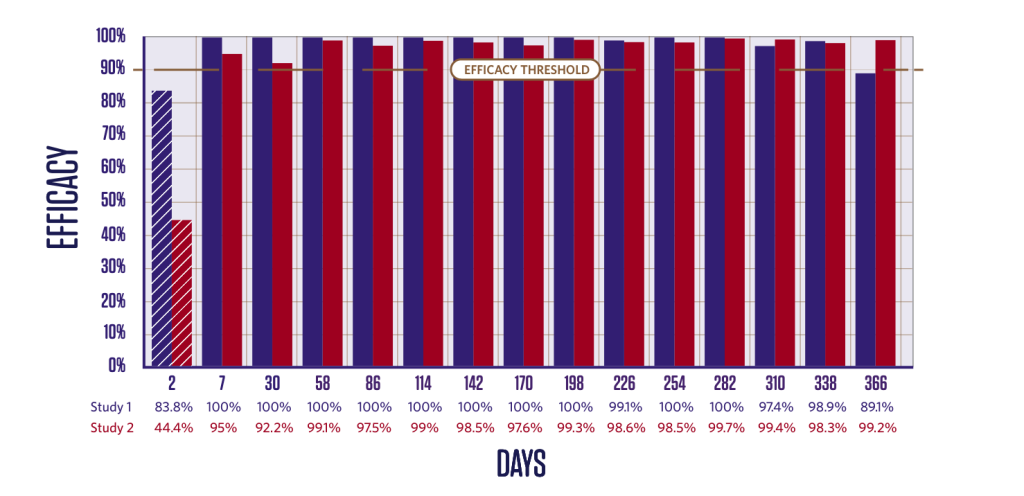
Persistent Ixodes ricinus killing activity from Day 3.
Newly-infested I. ricinus ticks are killed.3,6,7

2 groups (1 treated, 1 control);
10 dogs in each group.

Dogs were infested with 50 ticks on Day -2 and treated on Day 0. Efficacy against the initial infestation was evaluated on Day 2.
Persistent efficacy evaluated at intervals from Day 7 to Day 366 following repeated re-infestations.
*Tick counts are arithmetic means.


PRECISE DOSING

Multi-dose vial allows you to draw the exact dose you need for dogs of any size (15 mg fluralaner/kg body weight; 1ml/10kg body weight).

3 year shelf life of
BRAVECTO injectable,
3 months at room temperature after reconstitution.

For puppies less than 6 months of age, use BRAVECTO Chew or spot-on with a 12-week duration of efficacy against fleas and ticks*
*Please note BRAVECTO chewable tablets for dogs are indicated for immediate and persistent tick-killing activity for Rhipicephalus sanguineus for 8 weeks


HOW TO PREPARE FOR USE?
To ensure correct product preparation, follow the step-by-step mixing instructions carefully.
BRAVECTO INJECTABLE LIVE IN THE FIELD
Here is a video of Bravecto Injectable being administered into pets across the UK


What vets involved in clinical field studies say about Bravecto INJECTABLE
As a practice owner, I have been very engaged with Bravecto Injectable right from the start. The time-saving element for the practice is a significant advantage. I have 815 dogs on my Health Care Plan (HCP) and plan to move them all over at the time of their vaccine. This alignment reduces footfall, phone calls for the team and flea and tick consultations. So far, I have had 100% uptake from clients. It’s all in the delivery and how I tell the clients: ‘I have some good news for you. You know the chew you have to give every 12 weeks for fleas and ticks? Well, I can now give the same thing as an injection while you are here, and it lasts a whole year!’ Clients are over the moon, and every single one has moved over.
So far, I have used over 33 bottles of Bravecto injectable with no adverse events or dogs reacting badly to the 18-gauge needle. This approach also encourage sign-ups to the health care plan and my team is already engaged and fully onboard with this new product and process. With around 3000 clients, 80% of whom are dog owners, I plan to continue using Bravecto injectable to support my clients and their pets.
Neil Murdoch, BVMS MRCVS
Metro Vets
United Kingdom
It is a highly effective product, and to date, we had nothing on the market offering good antiparasitic (flea and tick) protection for a year.
Translated from original language: “original quotation”
Oscar Cortadellas, DVM PhD, Acred. AVEPA Internal Medicine
Clínica Veterinaria Germanias
Gandía, España
When I told the pet owners about the medication, they were excited and the question that was often asked was when it would be available.
Translated from original language: “original quotation”
Dr. Angela Friedrich
Tierarztpraxis Friedrich
Bad Neuenahr-Ahrweiler, Germany


ADDRESSING PET OWNER’S QUESTIONS ABOUT BRAVECTO INJECTABLE
Pet owners may have questions. Download our guide that helps answer commonly asked questions by pet owners who are considering using BRAVECTO injectable.
UNLEASH THE POWER OF BRAVECTO INJECTABLE

Experience a new unique formulation of fluralaner.
- 12 months of non-stop protection.
- Revolutionary crystal injectable formulation of fluralaner.
- Vet exclusive.
Putting administration back in the hands of the vets to ensure compliance and deliver convenience throughout the year.
FREQUENTLY ASKED QUESTIONS
Due to the possibility of fast-growing puppies outgrowing the dose over the 12 month duration, the product was developed for dogs and puppies 6-months of age and older.
There is no contraindication to concurrent vaccination. Vets should keep in mind vaccinations may cause fever, lethargy and inappetence, occurring within the first few days following vaccination.
Because the efficacy is sustained for an entire year following administration, dogs can be treated whenever they are unprotected against fleas and ticks. Dogs will not require another dose of flea and tick treatment for 12 months for the indicated parasites.
A margin of safety study was performed and demonstrated safety at doses of 1X (15 mg/kg), 3X (45 mg/kg) and 5X (75 mg/kg) administered every 4 months for a total of 6 doses. The only treatment-related finding was limited to injection site swellings that resolved over time.
The formulation is a suspension containing fluralaner powder and an 18 G needle is required to ensure the solid particles in suspension are accurately transferred from the vial into the syringe and from the syringe into the patient.
Dogs should be dosed 0.1 ml per kg of body weight. Dogs weighing 10 kg should be injected with 1.0 ml; dogs weighing 15 kg should be injected with 1.5 ml, etc.
To maintain efficacy of BRAVECTO injectable dogs should be re-dosed 12 months* after the previous injection.
The sterilization process used for the powder causes the glass to darken in colour, which is not the case for process used to sterilize the liquid vehicle.
After reconstitution, the suspension must be discarded 3 months’ from the date of reconstitution. Write the discard date on the label of the glass vial.
If the suspension is left to stand for more than 5 minutes then it should be shaken thoroughly for at least 30 seconds to resuspend the product before administration
This product does not require any special temperature storage conditions. After reconstitution, store below 30°C.
REQUEST FURTHER INFORMATION FROM THE MSD TEAM ON HOW TO INTRODUCE BRAVECTO INJECTABLE IN YOUR PRACTICE
We’ll answer your questions to help ensure you’re ready to prescribe BRAVECTO injectable and launch this product in style.
“*” indicates required fields


Discover our whole Bravecto family
REFERENCES
- MSD Animal Health UK, Study Summary REF-10145.
- MSD Animal Health UK, Study Summary REF-10157.
- BRAVECTO 150mg/ml powder and solvent for suspension for injection for dogs. Summary of Product Characteristics.
- MSD Animal Health UK, Study Summary REF-10166.
- MSD Animal Health UK, Study Summary REF-10142.
- MSD Animal Health UK, Study Summary REF-10143.
- MSD Animal Health UK, Study Summary REF-10161.
Bravecto® Spot-on for Cats and Bravecto® Chewable Tablets, Spot-on for Dogs and Bravecto® 150 mg/ml powder and solvent for suspension for injection for dogs contain fluralaner. POM-V.
Bravecto® Plus Spot-on for Cats contains fluralaner and moxidectin. POM-V.
Further information is available from the SPC, Datasheet or package leaflet.
Advice should be sought from the medicine prescriber. Prescription decisions are for the person issuing the prescription alone.
Use Medicines Responsibly.
MSD Animal Health UK Limited. Registered office Walton Manor, Walton, Milton Keynes MK7 7AJ, UK. Registered in England & Wales no. 946942.




